Disinfection Protocols for Biohazard Cleanup
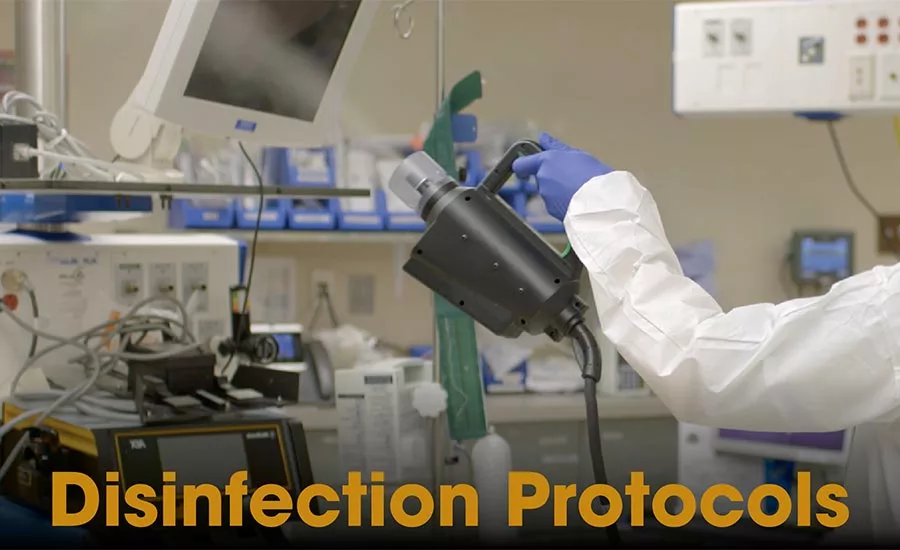
Regardless of whether you’re a longtime service provider or a novice in the industry, disinfection of dangerous pathogens in the healthcare industry can be a daunting task consisting of numerous variables. You find yourself having to factor in considerations such as: what critical pathogens need to be reduced in the surrounding environment? Are you prepared to treat a Candida auris outbreak? How quickly does the room need to be turned over? What rooms require disinfection, and do those rooms include electronic and delicate medical equipment? While these issues may seem overwhelming, it is prudent to answer every question in turn. Over time, they begin to get easier to address.
To help understand and navigate these considerations, the gravity of protocols must be understood. Protocols are essential in the healthcare industry. To create these protocols, infection preventionists work with the Environmental Services (EVS) team to reduce resistant pathogens levels including healthcare associated infections (HAIs) and reduce the spread of them. Creating these protocols includes implementing the best and most cutting-edge disinfection technologies to produce a successful room reduction in these resistant pathogens and HAIs. There are multiple disinfection technologies that healthcare facilities use, and it is important that the right questions are asked when selecting the solution. When seeking out the most suitable technology for a given facility, you need to consider facets of both the technology and the environment. Does the disinfectant have proven efficacy with a high-log reduction? Does the disinfectant kill the rapidly spreading drug-resistant fungus spore Candida auris? Does the product disinfect the area quickly? Does the disinfectant have a wet contact time? Does the disinfectant have to be wiped after the treatment? Does the disinfectant leave any residue? Can the disinfectant be used in many areas including rooms with sensitive materials and electronics? Most importantly, is the disinfectant EPA registered? These answers will ultimately determine which product will be used, and the outcome of that decision can have long-reaching effects on the facility, staff, and patients.
Having the proper disinfection and decontamination tools can make a world of difference when servicing a hospital, and one of the most significant aspects of a technology to consider can be versatility. More versatility means the ability to handle projects on a larger scope, and that can translate to reduced project overhead to better suit budgets. An EPA-registered technology, for example, can bring peace of mind with proven efficacy within categorized lists, such as List G for Norovirus, List K for Clostridium difficile, List L for Ebola Virus and List M for Avian Influenza (Flu) including H1N1, respectively. Additionally, scalability with no environmental HVAC adjustments means that you can reduce the TAT (Turnaround Time) of a hospital room turnover or their downtime. Versatility allows you to treat everything from waiting rooms to operating rooms – the same powerful technology that can be applied to the most sensitive of medical electronics can be just as effective on admission desks, nursing stations, physical therapy equipment, bathrooms and entryways. All of this means that, when the right questions are asked and answered, you are rewarded with a versatile no-touch enhanced technology that can find favor in several healthcare facilities such as hospitals, SNF’s, doctor’s offices and ambulatory surgery centers, or for control of general emergency outbreaks such as Ebola Virus, Clostridium difficile spores (C.diff) and the killer fungus Candida auris.

As a prime example of protocol development, a Delaware hospital found itself struggling with the transmission of hospital-acquired conditions, such as Clostridium difficile infections. This consequently placed patients and staff at risk with high rates of infection despite the offending rooms having previously been cleaned. With these emergency developments prompting the facility to act, the infection prevention team and environmental services department collaborated and asked those same questions, leading them to inquire about the use of SteraMist within their facility. With a proven track record of efficacy, EPA registration on lists G, K, L and M, handheld maneuverability, and innovative ionized Hydrogen Peroxide-based (iHP) technology, the facility settled on SteraMist to treat the emergency outbreak efficiently, and their goal was ultimately met. The dramatic reduction in their Clostridium difficile count has garnered them accolades from prominent names in the field of healthcare.
However, the facility didn’t simply stop at treating the emergency outbreak. The efficacy, ease-of-use, and non-corrosiveness of the process made it an invaluable tool beyond emergency remediation. Having rigorously proven itself, they began to implement the technology within their routine disinfection protocols. The new process allowed them to thoroughly treat high-touch areas and patient rooms, including items commonly found within such as hospital beds, monitors, call buttons, remote controls, guest chairs, floors, bathrooms and all electrical equipment. With limited departmental budgets in healthcare facilities worldwide, this kind of versatility can prove invaluable by not only maximizing return on investment, but also limiting liability and ensuring a pathogen free well-protected medical environment.
It should be noted that disinfection versatility is not only critical to those within the field of healthcare, but also to remediation specialists who are often called in to treat their facilities. The questions asked concerning in-house infection prevention is just as applicable when outsourcing treatment and can have a significant impact on the results of your investment. This is one of the driving ideas behind the creation of the TOMI Service Network (TSN), which aims to bring together a network of certified remediation specialists, all of whom are thoroughly trained in the deployment and utilization of SteraMist technology. The uniformity of the TSN network was designed to answer the crucial questions of infection prevention, including those concerning the reduction of critical pathogens, room turnover, and the impact of treatment on sensitive electronics. As a result, healthcare facilities can be left with guaranteed peace-of-mind while facility downtime is kept to a minimum when choosing a provider within the Network.

Despite the overwhelming considerations that may arise when approaching healthcare disinfection, each question ultimately has a basis in the protocols necessary to address a situation and the equipment used within them. Most facilities cannot afford to settle for less than the most powerfully advanced no-touch enhanced disinfection tools and being mindful of versatility and compatibility with sensitive electronics can minimize both short and long-term financial and litigation risk. To help navigate these considerations, it pays to surround yourself with other proven companies, protocols, and technology.
Looking for quick answers on restoration, remediation and cleaning topics?
Try Ask R&R, our new smart AI search tool.
Ask R&R →
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






